Barrett’s esophagus treatments focus on controlling acid reflux and stopping abnormal cells from turning into cancer. The right plan depends on how far the condition has progressed.
Table of Contents
ToggleBarrett’s esophagus happens when stomach acid repeatedly damages the esophagus lining. The cells change and start resembling intestinal cells, a process called intestinal metaplasia. About 3 to 5 percent of people with chronic acid reflux develop it. Most never get cancer, but the risk is real enough to manage actively.
How to Treat Barrett’s Esophagus
Treatments for Barrett’s esophagus depend on whether abnormal cells (called dysplasia) are present, how severe they are, and your overall health.
The treatment path breaks into three tracks:
- Control acid reflux to stop further damage
- Monitor the esophagus regularly with an endoscopy
- Remove or destroy abnormal cells if dysplasia appears
Most people with Barrett’s spend years on acid-suppressing medication plus regular monitoring. Treatment only becomes aggressive when dysplasia is found.
Guidelines for Barrett’s Esophagus Treatment
The guidelines for Barrett’s esophagus treatment come from two main sources: the American College of Gastroenterology (ACG) and the American Gastroenterological Association (AGA). Both follow stage-based management.
- No dysplasia: Take proton pump inhibitors (PPIs) daily. Get an endoscopy every 3 to 5 years.
- Low-grade dysplasia: Endoscopic ablation is now recommended, not just surveillance. The ACG updated this position in 2022 after studies showed ablation cuts cancer risk by up to 90% in low-grade cases.
- High-grade dysplasia: Treat immediately. Endoscopic mucosal resection (EMR) or radiofrequency ablation (RFA) is the standard. Surgery only enters the picture when endoscopic treatment fails or early cancer is already confirmed.
Treatment Based on Disease Stage
Barrett’s Without Dysplasia
No dysplasia means no precancerous cells yet. The tissue has changed, but it isn’t showing dangerous activity.
Treatment at this stage:
- PPIs (like omeprazole or pantoprazole) taken daily to reduce acid
- Endoscopy every 3 to 5 years
- Lifestyle modifications
PPIs in Barrett’s reduce the inflammation that drives cell mutation. Long-term PPI use in Barrett’s patients lowers the risk of progressing to dysplasia by approximately 71% compared to untreated patients.
Low-Grade Dysplasia
Cells are changing, but they’re still far from cancer. This stage used to be managed with watchful waiting alone. That approach has changed.
Current standard:
- Radiofrequency ablation (RFA) to destroy abnormal cells
- Follow-up endoscopy at 3 months, 6 months, then annually
- PPIs continue throughout the entire process
High-Grade Dysplasia
This is the last stage before cancer. Cells look significantly abnormal under a microscope.
Treatment:
- EMR for raised or nodular areas (removes a tissue sample for testing)
- RFA for flat areas of abnormal cells
- Cryotherapy as an alternative when RFA isn’t tolerated
- Repeat endoscopy every 3 months during the first year after treatment
Medications for Barrett’s Esophagus
PPIs form the backbone of Barrett’s esophagus treatments at every stage. Common options include omeprazole, esomeprazole, lansoprazole, and pantoprazole.
They block the pumps in your stomach that produce acid. Less acid means less damage to the esophagus lining.
PPIs alone don’t reverse Barrett’s. They stop it from getting worse. Think of them as a ceiling, not a cure.
Some doctors add H2 blockers like famotidine at night because acid spikes between 10 PM and 2 AM, even in patients already on PPIs. This overnight acid control is especially relevant for people with nocturnal reflux symptoms.
Long-term PPI use is generally safe, but research flags some concerns beyond 10 years of use, including mildly reduced magnesium absorption and a possible connection to chronic kidney disease. Doctors monitor for these in long-term patients.
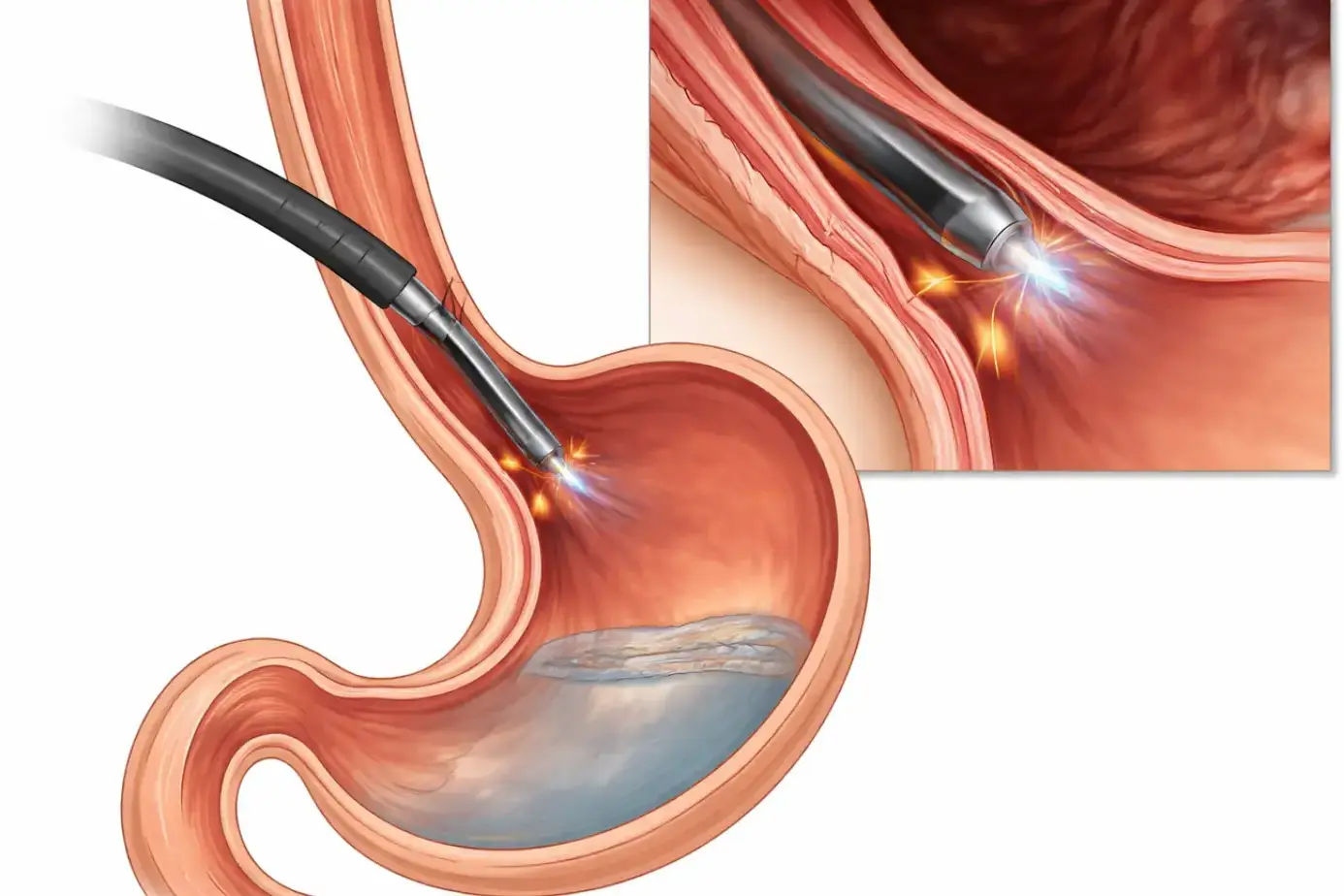
Endoscopic Treatments for Barrett’s Esophagus
All three procedures below are done through a flexible camera tube (endoscope) inserted through your mouth. No open surgery needed.
Radiofrequency Ablation (RFA): A small balloon with electrodes delivers controlled heat to the abnormal tissue. It burns away the top layer. Normal cells grow back. RFA achieves an 80 to 90% success rate for clearing dysplasia. Most patients need 2 to 3 sessions total.
Endoscopic Mucosal Resection (EMR): Used when a raised, irregular area needs to be removed and biopsied. The doctor injects fluid under the tissue to lift it, then cuts it off with a small snare. The removed piece goes to pathology to check for early cancer.
Cryotherapy: Extreme cold, either liquid nitrogen or CO2, freezes and kills abnormal cells. Used when RFA doesn’t work or when patients can’t tolerate thermal treatment. Success rates are slightly lower than RFA but still clinically meaningful.
Role of Hiatal Hernia in Barrett’s Treatment
A hiatal hernia forms when part of the stomach pushes up through the diaphragm into the chest. This weakens the valve between the stomach and esophagus, making acid reflux harder to control.
About 50 to 80% of Barrett’s patients have a hiatal hernia. When the hernia exceeds 3 cm, PPIs frequently fail to control acid adequately. In those cases, laparoscopic Nissen fundoplication surgically repairs the valve. Many patients reduce or stop PPIs entirely after surgery.
A 2019 study published in Gut found that Barrett’s patients with a repaired hiatal hernia showed slower disease progression than those managed on PPIs alone.
If Barrett’s isn’t responding well to medication, hernia evaluation is the logical next step, not a higher PPI dose.
Swallowing Difficulty and Barrett’s Esophagus Treatment
Swallowing difficulty during Barrett’s esophagus treatment becomes necessary when scar tissue, called a stricture, forms in the esophagus. This happens after RFA, EMR, or prolonged acid damage.
Symptoms include food getting stuck, pain when swallowing, or a persistent lump sensation in the throat.
Treatment options:
- Esophageal dilation: A balloon or dilator stretches the narrowed area during endoscopy. Most patients need 1 to 3 sessions.
- Steroid injections: Injected directly into the stricture during endoscopy to reduce scar tissue buildup.
- Dietary adjustment: Soft foods during flare-ups; avoid dry bread or tough meats until the area heals.
Strictures after RFA occur in approximately 5 to 12% of patients. They’re manageable, but early intervention produces better outcomes.
Lifestyle Changes That Support Treatment
Medication and procedures work better when supported by the right habits.
- Lose weight if your BMI is above 30. Abdominal fat increases internal pressure, pushing acid upward.
- Eat smaller meals. Large meals expand the stomach and temporarily weaken the lower esophageal sphincter.
- Stop eating at least 3 hours before lying down.
- Elevate the head of your bed by 6 to 8 inches using bed risers, not extra pillows.
- Cut coffee, alcohol, fatty foods, and citrus. These relax the valve that keeps acid in the stomach.
- Stop smoking. Smokers with Barrett’s progress to dysplasia faster than non-smokers in clinical studies.
Complications of Barrett’s Esophagus Treatment
The complications of Barrett’s esophagus treatment are real but manageable with proper follow-up.
After RFA:
- Chest discomfort lasting 1 to 3 days
- Stricture formation in 5 to 12% of patients
- Bleeding or perforation in less than 1% of cases
After EMR:
- Bleeding (occurs in roughly 2% of cases)
- Perforation (less than 0.5%)
- Strictures when a large tissue area is removed
Recurrence: Abnormal cells return in 20 to 25% of treated patients within 5 years. This is why follow-up endoscopy is not optional.
Can Barrett’s Esophagus Be Cured?
No. Barrett’s esophagus isn’t fully reversible. The goals of Barrett’s esophagus treatments are control and cancer prevention.
After successful ablation, normal-looking cells grow back. But the underlying genetic susceptibility that caused the original change remains. Acid damage can trigger abnormal cells again. Lifelong monitoring exists for exactly this reason.
When Surgery Is Considered
Surgery is not routine in Barrett’s esophagus treatments. It comes into consideration when:
- A large hiatal hernia makes acid control impossible with medication alone
- Endoscopic treatments fail to clear high-grade dysplasia
- Early-stage cancer is confirmed
The standard reflux surgery is laparoscopic Nissen fundoplication. If cancer is detected, partial or full esophagectomy becomes necessary depending on the stage.
Long-Term Monitoring and Follow-Up
Barrett’s requires lifelong follow-up. Even after successful treatment, regular endoscopy continues.
Standard schedule:
- After ablation: every 3 months in year one
- If results are clear: every 6 months in year two
- Then: annually for life
Missing surveillance is the biggest preventable risk. Esophageal cancer found at stage 1 carries a 5-year survival rate above 80%. Found at stage 4, it drops below 5%.
FAQs: Barrett’s Esophagus Treatments
What is the best treatment for Barrett’s esophagus?
For patients with dysplasia, radiofrequency ablation is the gold standard with 80 to 90% clearance rates. For Barrett’s esophagus treatments without dysplasia, daily PPIs combined with endoscopy every 3 to 5 years is the ACG-recommended approach.
Can Barrett’s esophagus be cured?
No. Barrett’s esophagus treatments reduce cancer risk and control the condition, but the original cellular change isn’t erased. Ablation allows normal cells to regrow, but genetic susceptibility remains. Lifelong monitoring is required regardless of treatment success.
Do all patients need surgery?
No. Surgery is reserved for patients with a large hiatal hernia, failed endoscopic treatment, or confirmed early esophageal cancer. The majority of Barrett’s patients manage long-term with PPIs and endoscopic procedures only.
What are common treatments?
Daily PPIs, radiofrequency ablation, endoscopic mucosal resection, and cryotherapy are the four most used Barrett’s esophagus treatments. The correct choice depends on whether dysplasia is present and its severity.
Can Barrett’s turn into cancer?
Yes, but slowly. About 0.3% of Barrett’s patients per year progress to esophageal adenocarcinoma. High-grade dysplasia carries a 6% annual cancer risk, which is why it requires immediate treatment rather than monitoring.
Is long-term treatment required?
Yes. Even after ablation successfully clears abnormal cells, PPIs and annual endoscopy continue for life. Recurrence happens in 20 to 25% of treated patients within 5 years.
What is dysplasia in Barrett’s esophagus?
Dysplasia means abnormal cell changes in the esophagus lining. Low-grade involves minor irregularities. High-grade shows significant abnormalities one step below cancer, with a 6% per year risk of progressing to esophageal adenocarcinoma without treatment.
How often is monitoring needed?
Without dysplasia: endoscopy every 3 to 5 years. After ablation: every 3 months in year one, every 6 months in year two, then annually. Monitoring frequency increases if dysplasia returns between scheduled appointments.
About The Author

Medically reviewed by Dr. Nivedita Pandey, MD, DM (Gastroenterology)
Dr. Nivedita Pandey is a U.S.-trained gastroenterologist and hepatologist with extensive experience in diagnosing and treating liver diseases and gastrointestinal disorders. She specializes in liver enzyme abnormalities, fatty liver disease, hepatitis, cirrhosis, and digestive health.
All content is reviewed for medical accuracy and aligned with current clinical guidelines.